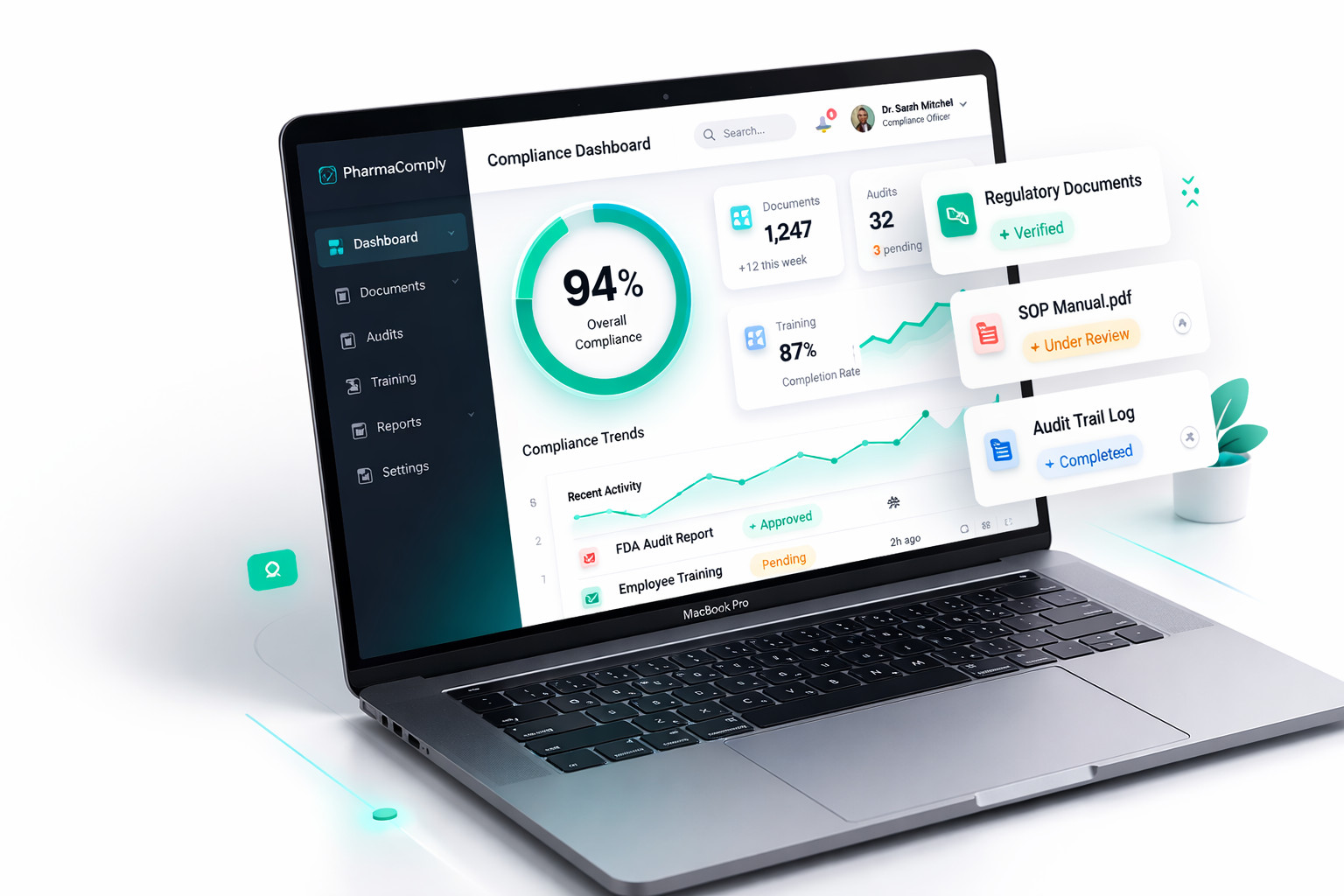
Live AI-Powered Pharma Document Management
Automate Your SOPs and
Stay Audit-Ready. Always.
DocSure uses AI agents to eliminate 80% of manual document management for pharmaceutical SMEs. Designed for Annex 11. GMP-ready. Built for European regulators.